What is Avogadro Number ?
Avogadro Number also known as Avogadro Constant is the number of atoms (6.022 x 1023) in one gram atomic weight (mole) of an element. Or also Avogadro Number can be defined as the number of molecules in a gram molecular weight (mole) of a compound.
It is sometimes given the symbol of NA or L and the unit of measure is mol-1 as per the International System of Units (SI).
Amadeo Avogadro
Avogadro Biography
Full name: Lorenzo Romano Amedeo Carlo Avogadro di Quaregna e di Cerreto, Count of Quaregna and Cerreto. Italian scientist born on the 9th of August 1776 in Turin, Sardinia. Deceased on the 9th of July 1856.
Avogadro 1837 – 1841 published 4 big volumes discussing in details the physics of matter.
Avogadro’s findings and in particular “Avogadro Number” were completely ignored until Stanislao Cannizarro in 1860 presented them at the Karlsruhe Conference. Which was four years after Avogadro left this world. The reason the conference was held is to clarify the confusion that existed at that time about atoms and molecules and their masses.
Still after that Cannizarro presented his findings, not all scientists were convinced. After a decade – with continued strong advocacy from Cannizarro – Avogardo’s hypothesis became more widely accepted and this is when it was called after Avogadro – Avogadro Number.
Today, Avogadro is considered one of the founders of atomic-molecular chemistry
Avogadro Number was first defined and introduced by Jean Baptiste Perrin as the number of atoms in one gram atomic weight of Hydrogen. Which basically means one gram of hydrogen. Later on, Avogadro Number had been redefined as the number of atoms in 12 grams atomic weight of the isotope Carbon-12 (12C). Furthermore, it also relates the amount of a substance to its molecular weight.
| Table shows the value of Avogadro Number  NA in different units |
|---|
| 6.022(74)x1023Â mol-1 |
| 2.731(12)x1026 (lb-mol)-1 |
| 1.7072(77)x1025 (oz-mol)-1 |
- Published in Water Chemistry, Water Treatment
What is Atomic Weight
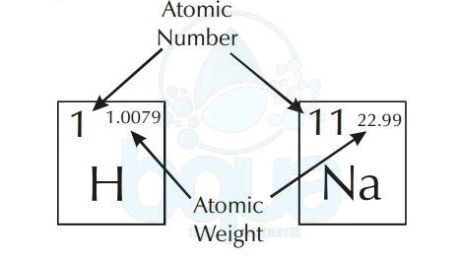
The atomic weight is simply the weight of one mole (6.022×10^23 atoms) of an element. The periodic table of elements shows the atomic weight on the upper right. The picture below shows an example of Hydrogen and Sodium atoms. Atomic weight of Hydrogen is 1.0079 when rounded to the nearest tenth = 1.0 gram. Same with Sodium atom, atomic weight is equal to 23.0 grams. For a molecule, to calculate the molecular weight we add the sum of atomic weights of the atoms in molecule.
atomic number and atomic weight in periodic table of elements
On the other hand, a gram atomic weight is the weight of one mole of an element, equal to its weight expressed in grams.
- Published in Water Chemistry, Water Treatment
Molecular Weight
A molecular weight is simply the weight of one mole (6.022×10^23 molecules) of a compound. Like the gram atomic weight, in the case of molecules, we describe the weight of one mole (6.022 x 10^23 molecules) as the gram molecular weight. To calculate the gram molecular weight of a molecule, we add the gram atomic weight of each atom comprising the molecule. For example, to calculate the gram molecular weight of water (H2O), we would add the gram atomic weight of oxygen plus two times the gram atomic weight of hydrogen.
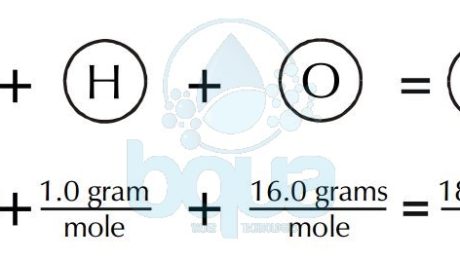
An example of H2O water molecule: The molecular weight is equal to the sum of atomic weights of Hydrogen and Oxygen atoms. Since there are two Hydrogen atoms and one Oxygen. Total is equal to 2(1.0 gram of Hydrogen) + 1(16.0 grams of Oxygen) = 2.0 + 16.0 = 18.0 grams molecular weight. It is also the weight of one mole of water.
Water Compound formed from Hydrogen Atoms and Oxygen Atoms
gram molecular weight of water molecule
Reverse Osmosis membranes will reject ions because of their charge. The membranes will also reject non-charged compounds (like organics) based upon molecular size. Generally, any organic less than 100 molecular weight will not be well rejected. Rejection of organics also depends upon the geometry of the molecule.
Molecular weight is important in organic chemistry since most organic molecules are quite large. Some polymers can have molecular weights in the millions.
- Published in Water Chemistry, Water Treatment
What is Periodic Table of Elements:
Atoms of different elements are distinguished only by the number of subatomic particles; electrons, protons and neutrons. Scientists decided to organize the elements in a periodic table based on the number of these particles in the atoms of each element. By the time, a chart was developed which described the atom structure of each element. This chart has come to be known as the periodic table of elements.
periodic table of elements including atomic number atomic weight
By observing the periodic table there are a lot of important information for each element. We see that each element in the periodic table has a Chemical Symbol which is usually the first one or two letters of the element’s name. For instance: H for Hydrogen, He for Helium, O for Oxygen, Ca for calcium, etc. In other cases, there are elements that have symbols which do not correspond to the name of the element. For instance: Na for sodium, K for potassium, etc. These letters are abbreviations of the Latin name of these elements. Na are the first two letters of Natrium (Sodium in Latin), K is the first letter of Kalium (Potassium in Latin) and so on.
Another observation in the periodic table is that each element has a corresponding number. This number is called the atomic number and it refers to the number of protons in the nucleus. Since we know that in neutral atoms (not charged) the number of protons is equal to the number of electrons, the atomic number also equals the number of electrons.
atomic number and atomic weight in periodic table of elements
The number which is greater than the atomic number is called the atomic weight. The atomic weight also called gram atomic weight is the weight of a particular number of atoms of the element in grams.
- Published in Water Chemistry, Water Treatment