Semipermeable Membrane Definition
Semipermeable membrane is a thin, soft, pliable sheet of material that allows certain substances to freely pass through it while restricting the passage of other substances. In the water treatment field, a semipermeable membrane allows water to pass freely while restricts the passing of dissolved materials.
Water passes through a semipermeable membrane into a solution of higher salt concentration. This phenomenon is called Osmosis. The semi-permeable membrane is a material through which water may pass, but prohibits the traveling of nearly everything else. Because of pressurized feed water enters each pressure vessel, a portion of the incoming water goes through the semipermeable membrane. The semi-permeable membrane selectively restricts the passage of suspended and dissolved elements in the raw water.
Semipermeable membrane separating TDS and TSSÂ
The incoming supply water and the dissolved and suspended substances which don’t pass throughout the semipermeable membrane go to waste. One stream enters an RO system and two streams exit. The permeate is the water stream and small amount of TDS which goes through the Semipermeable Reverse Osmosis membrane. While the concentrate is the waste stream containing the water and elements which don’t go through the semipermeable membrane. The concentrate is simply concentrated incoming stream of water. The concentrate contains the TDS and the TSS that are not allowable to pass through the semipermeable membrane.
Read more about the RO Membrane.
- Published in Technology, Water Treatment
What is Osmosis Definition
Osmosis is a natural process (phenomenon) that occurs in nature around us. The Osmosis process is even happening now within our bodies while reading this post.
A simple definition of Osmosis is that it is the tendency of a fluid to pass or flow through a semipermeable membrane into solution of higher concentration. Osmosis is the process whereby the water that we drink eventually ends up in our blood, muscles, and cells. The lining in our intestine is a membrane. The yolk of an egg has a membrane surrounding it that releases its contents when broken. If we simply wrap a piece of meat with plastic wrap, we can consider the plastic wrap a membrane.
A definition of permeable is when materials can pass through it. For example, a kitchen strainer or sieve is totally permeable to water. Meaning that water passes freely through it. Semipermeable means that some materials pass while others don’t. The kitchen strainer is an example. Since it allows water to pass freely through it but will not allow large solids to infiltrate.
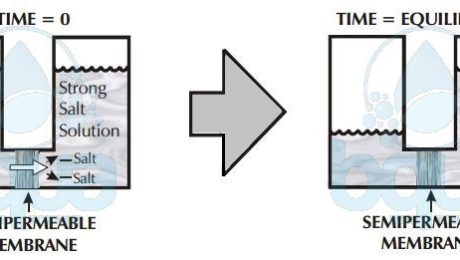
Back to Osmosis, water passes through semi-membrane from low salt concentration into a solution of high concentration. The reason is simple, let’s imagine having a salt solution and pure water separated by such a membrane. We know that water molecules may freely pass through the membrane but dissolved substances can’t. Given that all molecules are in motion, which liquid will have the greatest number of molecules colliding with the membrane? The pure water or the salt water?
Per given area of membrane, the pure clean water side will have more molecules of water that collide with the membrane than the side of water with salt. Water in Osmosis, therefore will pass from the side with pure or clean water to the salt water side. The higher the concentration of salt, the fewer the collisions of water molecules with the membrane. In Osmosis, water always passes through a semi-permeable membrane, therefore, from a lower salt concentrated side into a higher concentrated side.
osmosis: lower salt concentration to higher concentration
Osmosis then describes the tendency of fluid (water) to pass through a semi-permeable membrane, into a solution of higher concentration. The process stops when the number of water molecules colliding with either side of the membrane are equal. The number of collisions is a function of the salt concentration and the pressure on whichever membrane side. These are called osmotic pressure and applied pressure.
Osmosis reaches equilibrium when the concentration of dissolved solids at both compartments is equal. Meaning no more net flow from one side to another. The side that was the higher concentration solution however now has a higher water level than the other side due to osmosis. This difference in height between both sides is due to osmotic pressure.
Now that you learned about Osmosis, you might want to read about Reverse Osmosis (RO).
- Published in Water Chemistry, Water Treatment