What is Arsenic – Arsenic Element Definition
Arsenic concentrations in U.S. drinking waters are typically low. However, an estimated 5,000 community systems (out of 70,000) using groundwater and 370 systems (out of 6,000) using surface water were above 5 ug/L. These were primarily in the western states. Dissolution of arsenic-containing rocks and the smelting of nonferrous metal ores, especially copper, account for most of the arsenic in water supplies. Until the 1950s, arsenic element was also a major agricultural insecticide.
Arsenic element may be a trace dietary requirement and is present in many foods such as meat, fish, poultry, grain, and cereals. Market-basket surveys suggest that the daily adult intake of arsenic is about 50 ug, with about half coming from fish and shellfish. In fish, fruit, and vegetables, it is present in organic arsenical forms, which are less toxic than inorganic arsenic. However, arsenic is not currently considered essential (National Research Council, 1989). Extrapolating from animal studies, Uthus (1994) calculated a safe daily intake of between 12 and 40 ug.
In excessive amounts, arsenic element causes acute gastrointestinal damage and cardiac damage. Chronic doses can cause Blackfoot disease, a peripheral vascular disorder affecting the skin, resulting in the discoloration, cracking, and ulceration. Changes in peripheral nerve conduction have also been observed. Epidemiological studies in Chile, Argentina, Japan, and Taiwan have linked arsenic in drinking water with skin, bladder, and lung cancer (reviewed by Smith et al., 1992; Cantor, 1997). Some studies have also found increased kidney and liver cancer.
Ingestion of arsenical medicines and other arsenic exposures have also been associated with several internal cancers, but several small studies of communities in the United States with high arsenic levels have failed to demonstrate any health effects. Micronuclei in bladder cells are increased among those chronically ingesting arsenic in drinking water. Inorganic arsenate and arsenite forms have been shown to be mutagenic or genotoxic in several bacterial and mammalian cell test systems and have shown teratogenic potential in several mammalian species, but cancers have not been induced in laboratory animals.
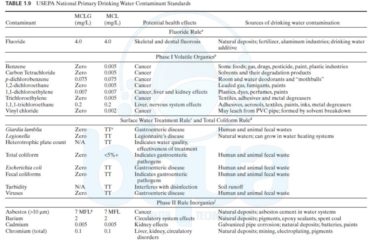
USEPA has classified arsenic as a human carcinogen, based primarily on skin cancer (U.S. Environmental Protection Agency, 1985).The ability of arsenic to cause internal cancers is still controversial. Under the NIPDWR regulations, an MCL of 50 ug/L had been set, but it is under review. Currently, USEPA’s Risk Assessment Council estimates that an RfD (for non-carcinogenic skin problems) ranges from 0.1 to 0.8 ug/kg/day−1, which translates into an MCLG of 0 to 23 ug/L. Based on a 1-in-10,000 risk of skin cancer, USEPA estimated that 2 ug/L might be an acceptable limit for arsenic in drinking water.