RO Membrane – Reverse Osmosis Membrane Materials, Types and Structures
Reverse osmosis RO membrane differs by the material of the membrane polymer and by structure and configuration. Based on its structure, RO membrane can be divided into two groups: conventional thin-film composite and thin-film nanocomposite. Based on the thin-film material, conventional reverse osmosis RO membrane at present is classified into two main groups: polyamide and cellulose acetate. Depending on the configuration of the membrane within the actual membrane elements (modules), the reverse osmosis membrane materials is divided into three main groups: spiral-wound, hollow-fiber, and flat-sheet (plate-and-frame).
Conventional Thin-Film Composite Membrane Structure
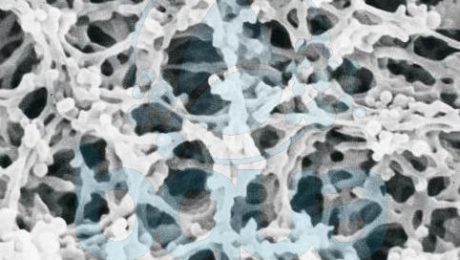
The reverse osmosis RO membrane most widely used for desalination at present are composed of a semipermeable thin film (0.2 um), made of either aromatic polyamide (PA) or cellulose acetate (CA), which is supported by a 0.025- to 0.050-mm microporous layer that in turn is cast on a layer of reinforcing fabric (Fig. 1.1 for a membrane with an ultrathin PA film). The 0.2-um ultrathin polymeric film is the feature that gives the RO membrane its salt rejection abilities and characteristics. The main functions of the two support layers underneath the thin film are to reinforce the reverse osmosis membrane structure and to maintain membrane integrity and durability.
Fig 1.1: Structure of a typical reverse osmosis RO membrane
The dense semipermeable polymer film is of a random molecular structure (matrix) that does not have pores. Water molecules are transported through the membrane film by diffusion and travel on a multidimensional curvilinear path within the randomly structured molecular polymer film matrix. While the thin-film RO membrane with conventional random matrix-based structure shown in Fig. 1.1 is the type of membrane that dominates the desalination industry, new thin-film membrane of more permeable structure are currently under development in research centers worldwide.
Thin-Film Nanocomposite RO Membrane Structure
Thin-Film Nanocomposite TFC membrane either incorporate inorganic nanoparticles within the traditional membrane polymeric film structure (Fig. 1.2) or are made of highly structured porous film consisting of a densely packed array of nanotubes (Fig. 1.3). In Fig. 1.2, part A shows the thin film of a conventional PA membrane, supported by the polysulfone support layer. Part B shows the same type of membrane with embedded nanoparticles (labeled “NP”).
Fig 1.2: Polyamide reverse osmosis RO membrane with nanoparticles
Fig 1.3: Reverse osmosis RO membrane with carbon nanotubes
Nanocomposite reverse osmosis membrane material reportedly has higher specific permeability than conventional RO membrane at comparable salt rejection. Which is the ability to transport more water through the same surface area at the same applied pressure). In addition, thin-film nanocomposite membrane have comparable or lower fouling rates in comparison to conventional thin-film composite RO membrane operating at the same conditions. And they can be designed for enhanced rejection selectivity of specific ions. If membrane material science evolved to a point where the membrane structure could be made of tubes of completely uniform size, theoretically the membrane could produce up to 20 times more water per unit surface area than the RO membrane commercially available on the market today. As membrane material science evolves toward the development of membrane with more uniform structure, the further development of RO desalination membrane technology has the potential to yield measurable savings in terms of water production costs.
Cellulose Acetate CA Membrane
The thin semipermeable film of the first membrane element – developed in the late 1950s at the University of California, Los Angeles – was made of cellulose acetate (CA) polymer. While CA membrane has a three-layer structure similar to that of PA membrane, the main structural difference is that the top two layers (the ultrathin film and the microporous polymeric support) are made of different forms of the same CA polymer. In PA membrane these two layers are of completely different polymers – the thin semipermeable film’s made of polyamide, while the microporous support’s made of polysulfone (see Fig. 1.1). Similar to PA membrane, CA membrane has a film layer that is typically about 0.2 um thick; but the thickness of the entire membrane (about 100 um) is less than that of a PA membrane (about 160 um).
One important benefit of CA membrane is that the surface has very little charge and is considered practically uncharged, as compared to PA membrane, which have negative charge and can be more easily fouled with cationic polymers if such polymers are used for source water pretreatment. In addition, a CA membrane have a smoother surface than the PA membrane, which also renders them less susceptible to fouling.
CA membrane has a number of limitations, including the ability to perform only within a narrow pH range of 4 to 6 and at temperatures below 35°C (95°F). Operation outside of this pH range results in accelerated membrane hydrolysis, while exposure to temperatures above 40°C (104°F) causes membrane compaction and failure. Significant use of acid for normal plant operation requires reverse osmosis RO permeate adjustment by adding a base (typically sodium hydroxide) to achieve adequate boron rejection; in order to maintain the RO concentrate pH below 6, the pH of the feed water to the CA membrane has to be reduced to between 5 and 5.5.
CA membrane experiences accelerated deterioration in the presence of microorganisms capable of producing cellulose enzymes and bioassimilating the membrane material. However, they can tolerate exposure to free chlorine concentration of up to 1.0 mg/L. Which helps to decrease the rate of membrane integrity loss due to destruction by microbial activity. Since CA membrane has a higher density than PA membrane, it creates a higher headloss when the water flows through the membrane. Therefore they have to be operated at higher feed pressures, which results in elevated energy expenditures. CA membrane is used in municipal applications for saline waters with very high fouling potential (mainly in the Middle East and Japan) and for ultrapure water production in pharmaceutical and semiconductor industries. That is despite their disadvantages and mainly because of their high tolerance to oxidants (chlorine, peroxide, etc.) as compared to PA membrane.
Aromatic Polyamide Membrane
Aromatic polyamide (PA) membrane is the most widely used type of RO membrane at present. They have found numerous applications in both potable and industrial water production. The thin polyamide film of this type of semipermeable membrane is formed on the surface of the microporous polysulfone support layer (Fig. 1.1). It is formed by interfacial polymerization of monomers containing polyamine and immersed in solvent containing a reactant to form a highly cross-linked thin film. PA membrane operates at lower pressures and have higher productivity (specific flux) and lower salt passage than CA membrane. Which are the main reasons they have found a wider application at present.
While CA membrane has a neutral charge, PA membrane has a negative charge when the pH is greater than 5. Which amplifies co-ion repulsion and results in higher overall salt rejection. However, when pH < 4, the charge of PA membrane changes to positive and rejection reduces significantly to lower than that of a CA membrane. Another key advantage of PA membrane is that they can operate effectively in a much wider pH range (2-12). This allows easier maintenance and cleaning. In addition, PA membrane is not biodegradable and usually have a longer useful life – 5-7 years versus 3-5 years. Aromatic polyamide membrane is used to produce membrane elements for brackish water and seawater desalination, and nanofiltration.
Comparison between PA and CA Membrane
It should be noted that PA reverse osmosis membrane material is highly susceptible to degradation by oxidation of chlorine and other strong oxidants. For example, exposure to chlorine longer than 1000 mg/L-hour can cause permanent damage of the thin-film structure and can significantly and irreversibly reduce membrane performance in terms of salt rejection. Oxidants are widely used for biofouling control with RO and nanofiltration membranes. Therefore, the feed water to PA membrane has to be dechlorinated prior to separation. Table 1.4 below presents a comparison of key parameters of polyamide and cellulose acetate RO membrane in terms of their sensitivity to feed water quality.
| Parameter | Polyamide Membrane PA | Cellulose Acetate CA Membrane |
| Salt rejection | High (> 99.5%) | Lower (up to 95%) |
| Feed pressure | Lower (by 30 to 50%) | High |
| Surface charge | Negative (limits use of cationic pretreatment coagulants) |
Neutral (no limitations on pretreatment coagulants) |
| Chlorine tolerance | Poor (up to 1000 mg/L-hours); feed de-chlorination needed |
Good; continuous feed of 1 to 2 mg/L of chlorine is acceptable |
| Maximum temperature of source water | High (40 to 45°C; 104 to 113°F) | Relatively low (30 to 35°C; 86 to 95°F) |
| Cleaning frequency | High (weeks to months) | Lower (months to years) |
| Pretreatment requirements | High (SDI < 4) | Lower (SDI < 5) |
| Salt, silica, and organics removal | High | Relatively low |
| Biogrowth on membrane surface | May cause performance problems | Limited; not a cause of performance problems |
| pH tolerance | High (2 to 12) | Limited (4 to 6) |
Table 1.4: Comparison between Polyamide PA Membrane and Cellulose Acetate CA Membrane materials
Polyamide PA membrane is the choice for most RO membrane installations today. Mainly because of their higher membrane rejection and lower operating pressures. Exceptions are applications in the Middle East, where the source water is rich in organics. Thus cellulose acetate membrane offers benefits in terms of limited membrane biofouling and reduced cleaning and pretreatment needs. CA membrane provides an acceptable tradeoff between lower fouling rates and chemical cleaning costs. Also higher operating pressures and power demand on the other. Because of the relatively lower unit power costs in the Middle East. There are newer generations of lower-fouling PA membranes today on the market. The use of CA membrane elements is likely to diminish in the future.
- Published in Seawater Desalination, Technology, Water Treatment
What is a Cellulose Acetate Membrane
The thin semi-permeable film of the first Reverse Osmosis membranes was developed in the late 1950s at UCLA (University of California Los Angeles). It was made of cellulose acetate (CA) polymer. A Cellulose Acetate membrane have a three-layer structure similar to that of a Polyamide Thin Film Composite TFC membrane. The main structural difference is that the top two layers (the ultrathin film and the microporous polymeric support) are made of different forms of the same Cellulose Actetate polymer.
Cellulose is a polymer that is made up of repeating units (monomers) of C6H10O5. (Note: A monomer is a molecule which comes together with other identical monomers to form a chain of monomers, called a polymer). The number of acetates on the cellulose molecules affects the semi-permeability and other characteristics of the membrane. In general, the following are some of the most important differences between diacetate and triacetate membranes.
cellulose polymer cellulose acetate membrane
In a TFC membrane these two layers are made of completely different polymers. The thin semi-permeable film is polyamide, while the microporous support is polysulfone. Similar to a TFC membrane, a Cellulose Acetate membrane have a film layer that is typically about 0.2 um thick. But the thickness of the entire membrane (about 100 um) is less than that of a TFC membrane (about 160 um). One important benefit of a Cellulose Acetate membrane is that the surface has very little charge and is practically uncharged. As compared to a TFC membrane, which have a negative charge and can be more easily fouled with cationic polymers. If such polymers are used for source water pre-treatment.
cellulose acetate membrane pores under microscope
In addition, a Cellulose Acetate membrane have a smoother surface than a TFC membrane. Which also renders them less susceptible to fouling. Cellulose Acetate membrane have a number of limitations. Including the ability to perform only within a narrow pH range of 4 to 6 and at temperatures below 35 C (95 F). Operation outside of this pH range results in accelerated membrane hydrolysis, while exposure to temperatures above 40 C (104 F) causes membrane compaction and failure. In order to maintain the Reverse Osmosis concentrate pH below 6, the pH of the feed water to the cellulose acetate membrane are reduced to between 5 and 5.5. This results in significant use of acid for normal plant operation and requires Reverse Osmosis permeate adjustment by addition of a base (typically sodium hydroxide) to achieve adequate boron rejection.
Cellulose Acetate membrane experience accelerated deterioration in the presence of microorganisms. Since they’re capable of producing cellulose enzymes and bioassimilating the membrane material. However, they can tolerate exposure to free chlorine concentration of up to 1.0 mg/L. Which helps to decrease the rate of membrane integrity loss due to destruction by microbial activity.
Since Cellulose Acetate membrane have a higher density than a Polyamide TFC membrane. They create a higher headloss when the water flows through the membranes. Therefore a cellulose acetate membrane operates at higher feed pressures, which results in elevated energy expenditures. Despite their disadvantages, cellulose acetate membrane have high tolerance to oxidants (chlorine, peroxide, etc.). As compared to a PA TFC membrane. Cellulose Acetate membrane are used in municipal applications for saline waters with very high fouling potential. Mainly used in the Middle East and Japan in seawater reverse osmosis plants, and for ultrapure water production in pharmaceutical and semiconductor industries.
- Published in Technology, Water Treatment