What is a Covalent Bond
Atoms can bond by sharing electrons in their outermost orbitals and thus giving them the privilege of having a full outermost orbital. For example, the element Hydrogen exists as H2 molecule consisting of two atoms of Hydrogen atom. A covalent bond is simply the force between two atoms resulting from the sharing of electrons in the outermost orbital. Therefore a covalent bond attracts two atoms very close to each others because both atoms share the same electrons in their outermost orbitals.
hydrogen gas formed by covalent bond sharing electron between two hydrogen atoms
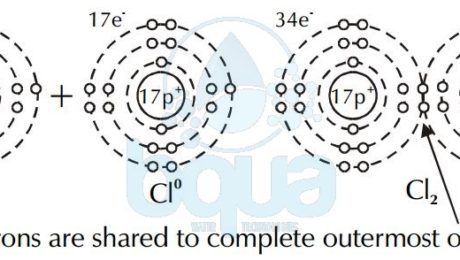
The case of Hydrogen cannot be an ionic bond. That is because Hydrogen ions each have the same charge and therefore are not attracted to one another. This is why a covalent bond happens only when two atoms have a difference in electronegativity of > or = 0.9. Instead, electrons are shared between the atoms to fill the outermost orbital. This type of bond is called a covalent bond. Two atoms of chlorine (Cl0) share a pair of electrons to form chlorine gas (Cl2).
electron shared covalent bond chlorine atom forming chlorine gas
Some molecules formed by covalent bonds may still give up or acquire one or more electrons resulting in a net positive or negative charge. These molecules are called molecular ions.
A covalent bond is very strong. The energy needed to break it is bigger than the thermal energy existing at 25°C which is room temperature. The thermal energy at 25°C is <1 kCal/mole kilo calorie per mole, while to break a typical Carbon covalent bond in an Ethane molecule we need about 83 Kilocalorie per mole.
Properties of molecules defined by covalent bond
While a central atom in a molecule attracts other atoms by covalent bonds, the bonds between these atoms are forming particular angles between them. The force of repulsion among these atoms and particularly the outermost electrons is what shape and determine the angles degrees. The angles and the shape formed is what gives the molecule its properties and shape. An example is the angle formed between Oxygen and Hydrogen atoms in a water molecule, an angle of 105°. You can read about the molecular geometry.
When atoms formed in a covalent bond have the same or very close electronegativity – typically less than 0.9 – the bond (the force of attraction) is equal between electrons since they’re almost identical. This kind of bond is called a non-polar bond. Carbon-Carbon and Carbon-Hydrogen bonds (also called Hydrocarbon) is an example on non-polar bonds.
A polar bond occurs when the difference in electronegativity between two atoms are more than or equal to 0.9. In a polar bond, there’s a partial negative charge (δ−) and a partial positive charge (δ+). This is the case in a water molecule H2O. The bond between Oxygen and Hydrogen is polar since difference in electronegativity is 1.2. (Electronegativity of Oxygen is 3.4 and Electronegativity of Hydrogen is 2.2).
- Published in Water Chemistry, Water Treatment
What is an Atom Definition
An atom is the most discrete indivisible particle that forms an element. Atoms of the same element are identical; different elements have non-similar atoms. The most simple of all elements is Hydrogen.Â
Example: Hydrogen atoms are similar while Oxygen atoms are different than Hydrogen atoms. Two hydrogen atoms and one Oxygen combined form the water molecule H2O.
The atoms are extremely small. Therefore, even the most powerful microscopes cannot see them. Actually, 6.022X10^23 hydrogen atoms (Avogadro Number) weigh only one gram. There are 454 grams in one pound.
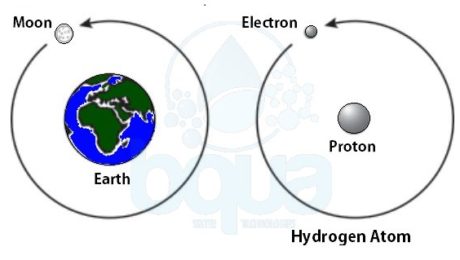
In order to understand the nature of the atom, we should identify the forces that are holding it together. Much as our plant, gravity is the force holding the moon in its orbit around the earth. Gravity is the force of attraction between two large masses.
In the case of the atom, the small size of the particles and the speed at which the electron travels would prohibit a force such as gravity from being able to hold the atom together. Scientists have learned that electrostatic force is the force that maintains the atom.
Atoms Composition
While atoms are composed of small subatomic particles; these particles are the electron, the proton and the neutron. The proton and neutron are found in the nucleus which is the center of the atom. The electron is negatively charged and orbits around the nucleus.
atom composed of subatomic particles electron proton neutron
Atoms from different elements are constituted of the same type of subatomic particles. However, the proportions of the subatomic particles are different for each element. Hydrogen for example has two subatomic particles; an electron and a proton. In the case of hydrogen, a single electron orbits a single proton. ÙAn example is the moon which orbits the earth. Same as in the case of earth, the proton of the hydrogen atom is much larger than the electron.
an electron orbits proton in nucleus in an atom like moon orbits earth
Moreover, if we add a subatomic particle to a Hydrogen atom, we create another atom of another element. Note: The number of subatomic particles is what characterizes atoms of different elements. In addition, if we want to create a Helium atom, we should only add an additional electron, an additional proton and two neutrons to the Hydrogen atom. As a result, we’ll have two electrons orbiting four other particles, two protons and two neutrons in the nucleus.
- Published in Water Chemistry, Water Treatment